Our Solution
Providing digital data capture and review across quality and production.
Supporting real-time insight and longer-term continuous improvement.
Capture - Analyze - React
Using a specialized real-time, digital data capture system improves the accuracy and timeliness of data collection; a key step in eliminating waste, reducing quality losses and supporting continual process improvement.
With QIS, you can connect all aspects of production; process data, quality data, documentation, and materials tracking, from suppliers through to customers. This is all supported with in-built analytical tools, exceptional reporting, and the capability to build complex certificates of analysis.
QIS has been proven to reduce costs, improve productivity and most importantly ensure the quality and safety of your products. Wherever data is captured from it is instantly visible and presented across the whole organization for immediate investigation, analysis, and reporting.

Key Features
Quality Control & Assurance
Ensure product quality and consistency in real-time
Statistical Process
Control (SPC)
Reduce waste, rework and raw material usage
Real-time Process Monitoring
Identify and respond to
changes in your process
OEE - Production Tracking
Prevent downtime, performance and quality losses
Reporting & Analysis
Crunch months of quality, process and production data in seconds
Materials Tracking
& Traceability
Stop substandard materials entering your process
Automated Data Capture
Communicate and exchange data with 3rd party systems and applications
Digital Laboratory
Manage data, assets, calibration schedules and Gage R&R
Document Management
Confirm conformance to standards and procedures
Ensure process and product quality - from supplier to customer and everything in between
Goods in / Vehicle Inspection
Raw material testing
Material Preparation
Cleaning
Production
Steps
Pre-production
Packaging
Laboratory
Final Inspection

Quality Control
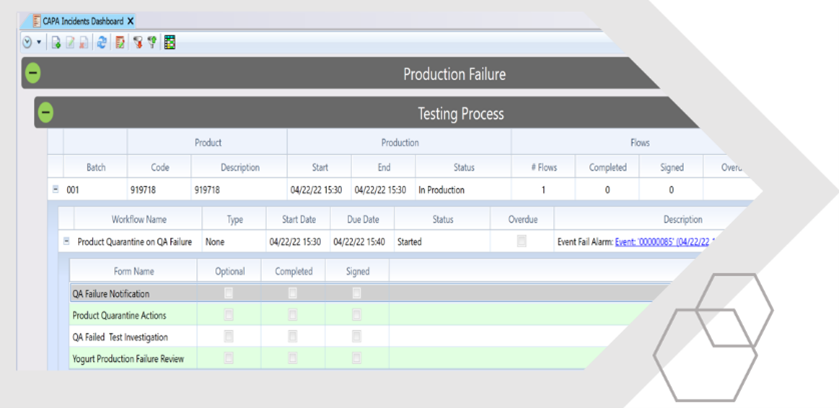
Facilitating the digital collection, analysis and presentation of production, quality and laboratory data, QIS gives paperless access to real-time, intelligence across your manufacturing operation.
Allowing you to control what information gets collected and exactly when it needs to be recorded, ensures all checks and tests are completed on time, every time. The ‘single window’ view enables teams to correct problems quickly and learn the cause and effect of actions taken.
Statistical Process Control
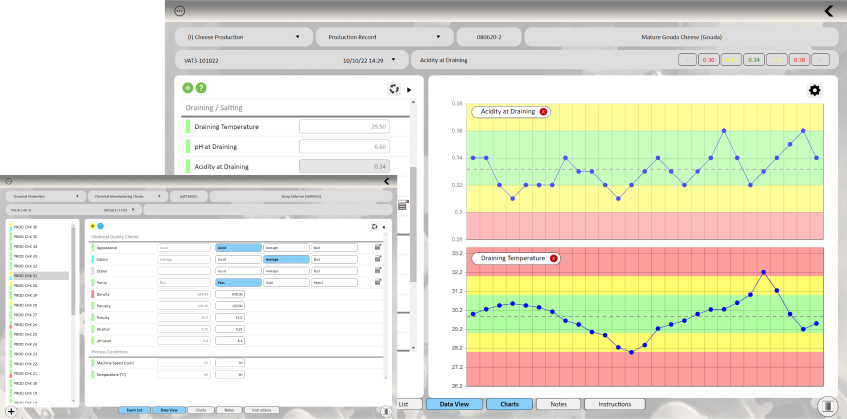
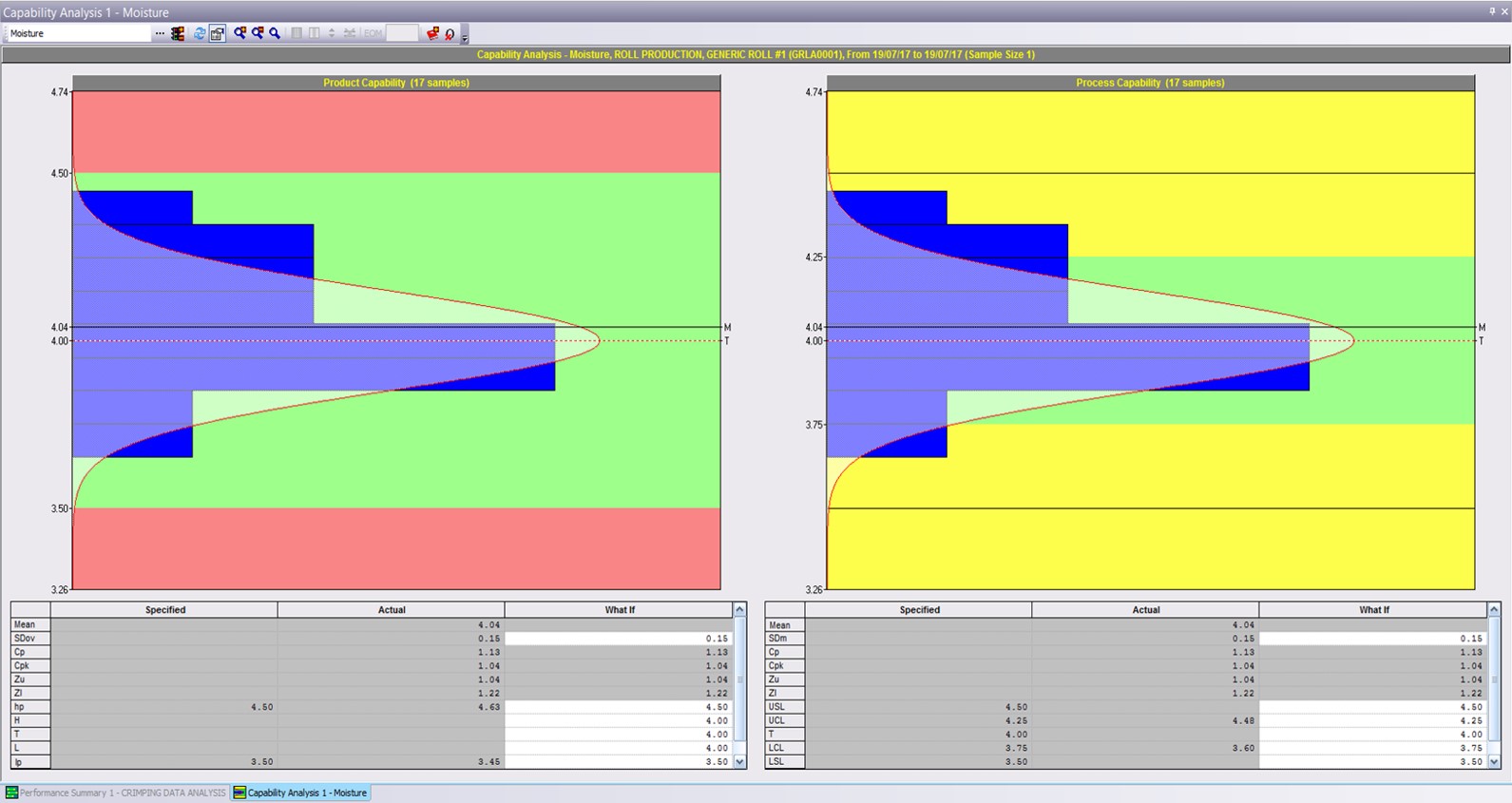
QIS is founded on the principals of Statistical Process Control, helping transform your data into actionable intelligence, supporting fact-based continuous improvement decisions.
QIS allows you to easily collate, visualize and analyze your data in seconds. Real-time charts, provide instant visibility of trends and relationships between process conditions and quality output, whilst powerful but easy to use statistical tools enable you to drill deep into your data to confirm capability and adherence to specification.

Real-time Process Monitoring
Capture rapidly changing time-based data outside of the standard event-based structure
Ensure critical process parameters are met throughout a production event
Scrutinize process conditions aligned to quality and performance

Reporting & Analysis
Get quick and easy access to real-time and historical data with a comprehensive selection of dashboards, in-built analysis tools and flexible
reporting options. All the analysis tools in QIS are designed to be quick and simple to use, and readily accessible for review on-line, empowering operators and managers to make faster and more accurate decisions – when it matters. Crunch months and years of data in seconds and review within the in-built analytical modules or
make available for reporting.
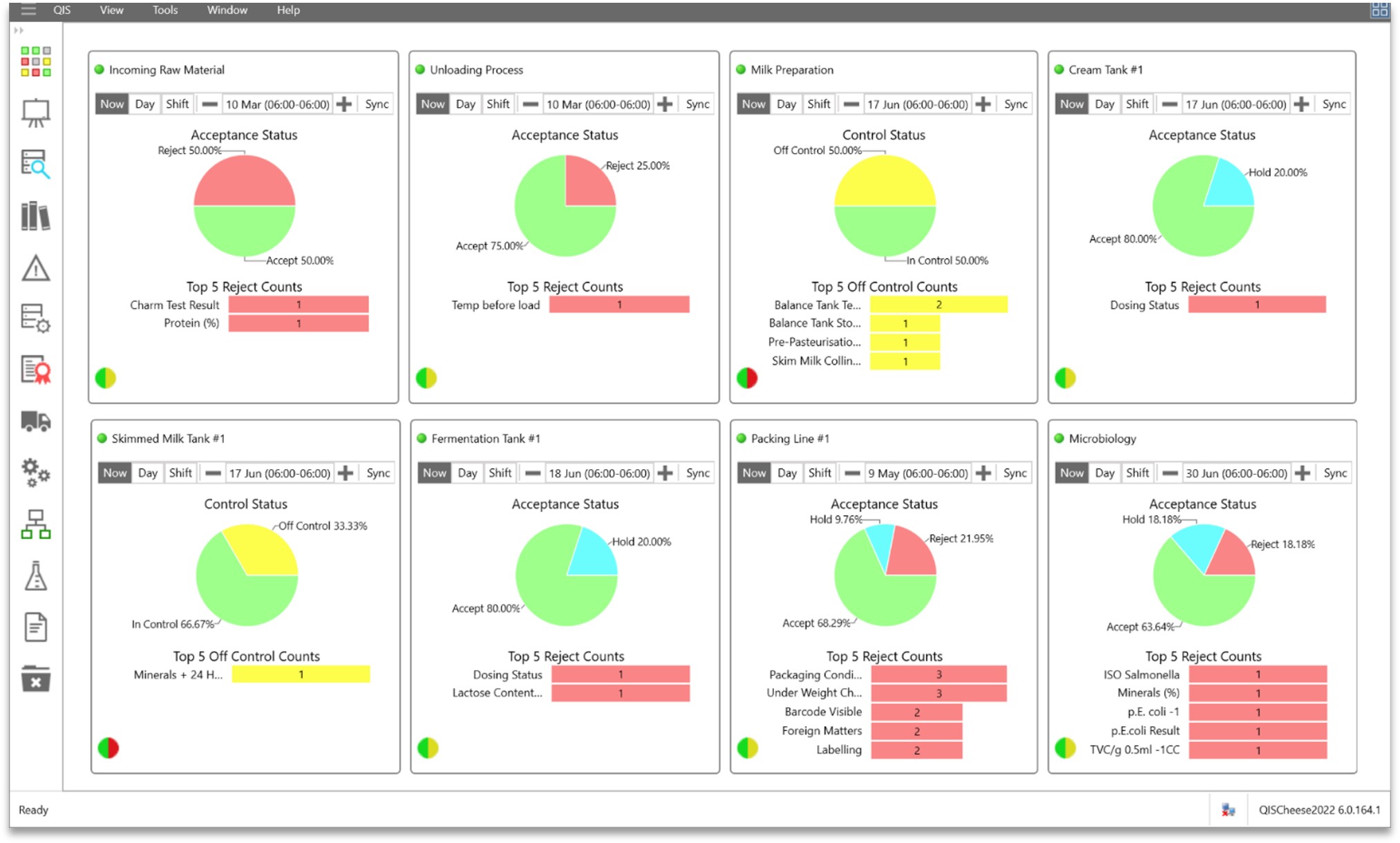
Real-time, customizable and interactive dashboards provide quick access to intelligence from across your production processes.
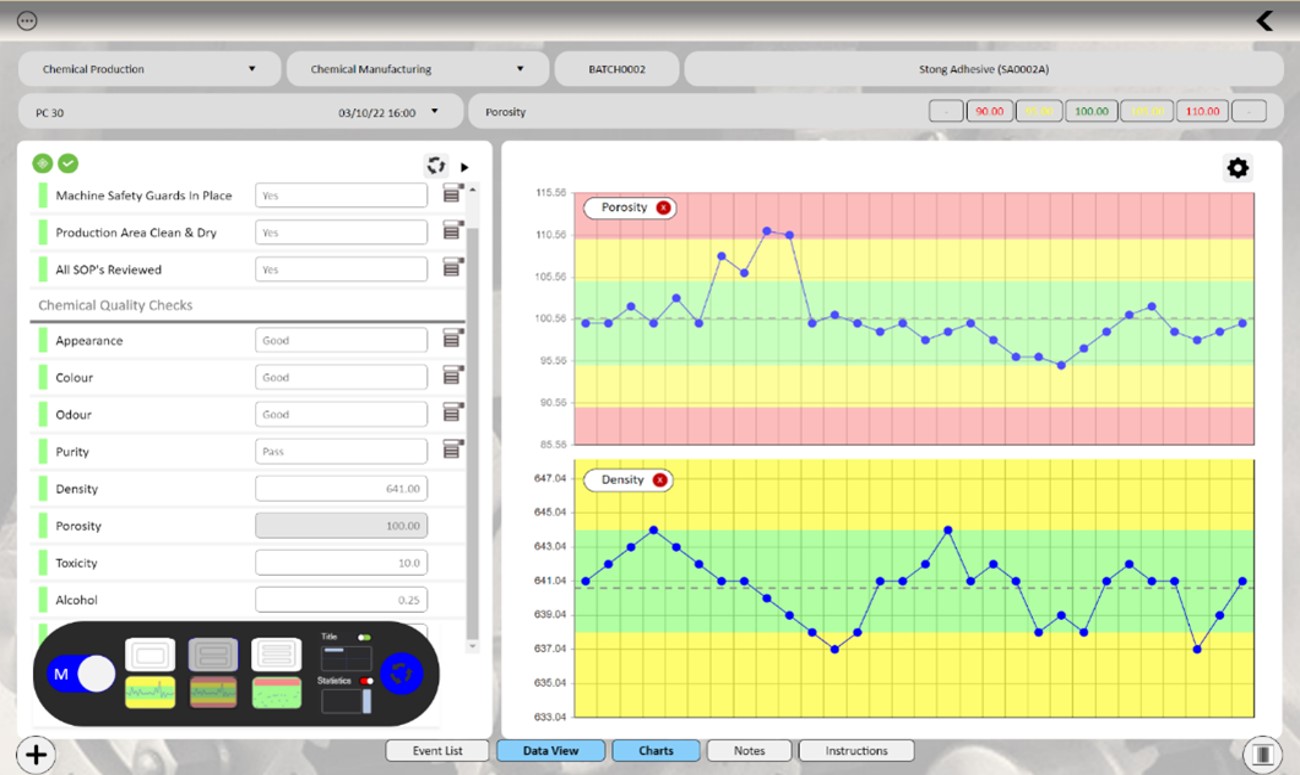
Instant charting allows operators to distinguish between random
fluctuations and true shifts within the data.
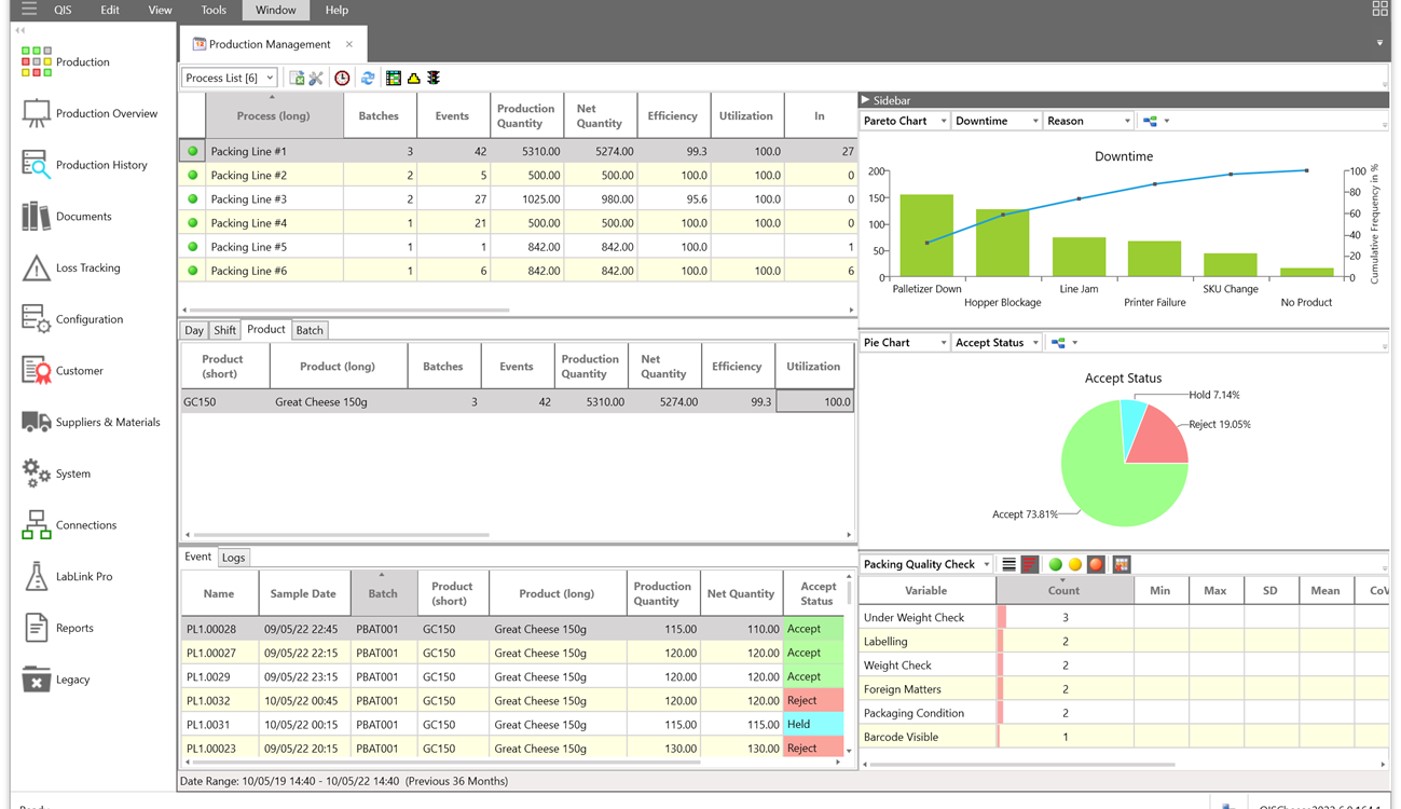
Rapid review of capability helps establish effective control limits and drive confidence in the ability to meet customer requirements.
Certificates of Analysis
A Certificate of Analysis assures your customer that the properties of the products you have supplied meet specification. This makes accuracy of the content critical and its presentation important. It may also be the last opportunity you have for quality assurance before a shipment is made.

Document Management
Document viewer displays your document along side the relevant data in the QIS logsheet
Manage both manufacturing and customer specifications to drive acceptance statuses
Strict change approval rights and revision history management


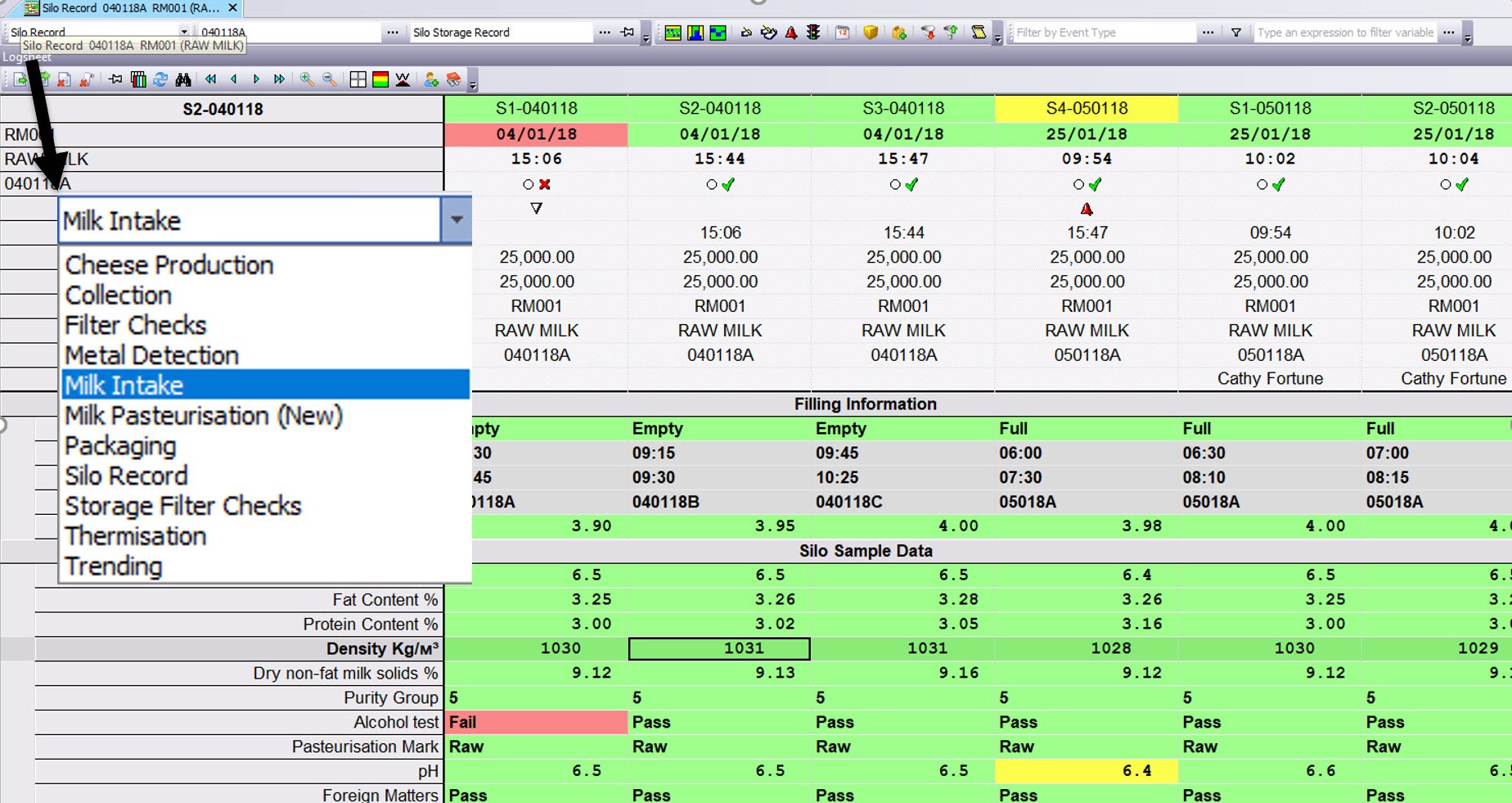
Digital Laboratory
Enabling laboratory technicians to automate, retrieve and manage laboratory test
data and provide accurate reporting of this to the production floor. From sample testing and quality results, to sharing of test methods and complete asset management.
As checks are completed in the Laboratory, QIS provides instant confirmation and visual feedback to both Lab Technicians and Operators on the line.
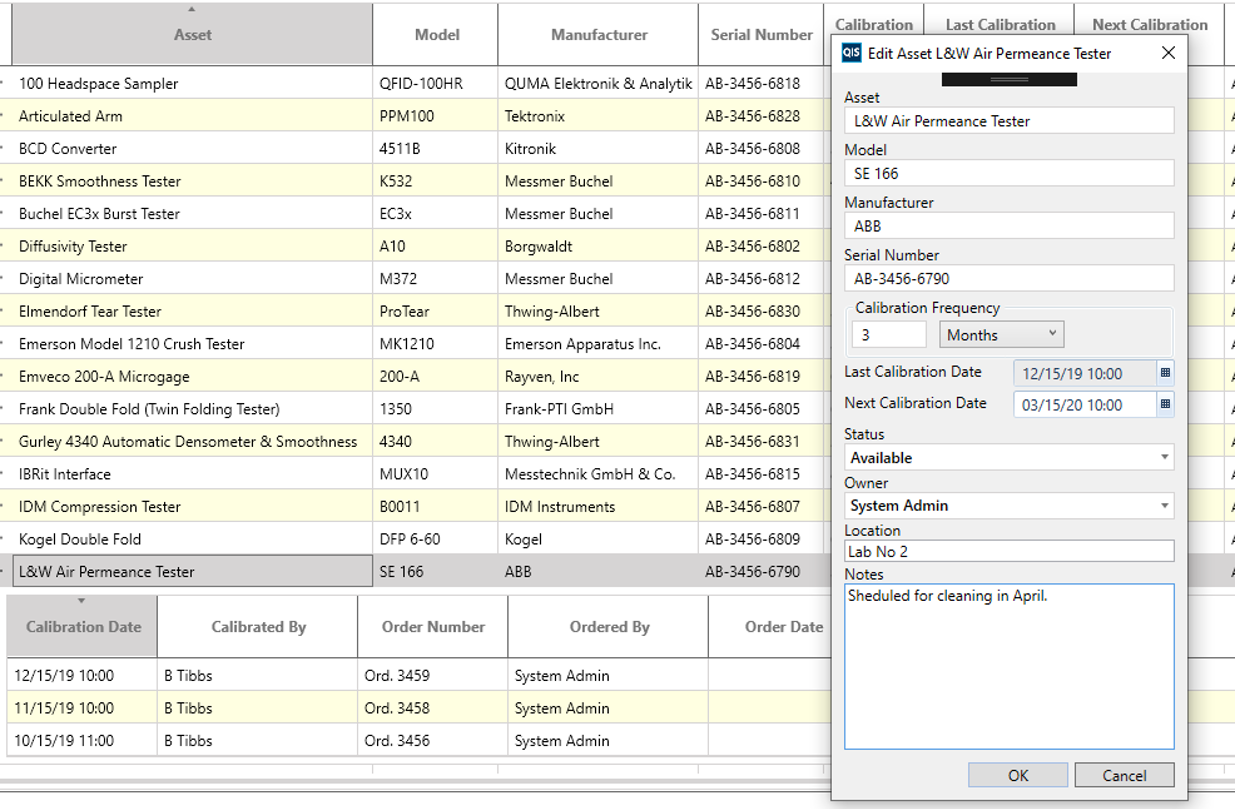
Manage all instrument assets, record their pertinent details and log and track calibration requirements. Then utilize the calibration calendar to track all calibration events and subscribe to notifications.
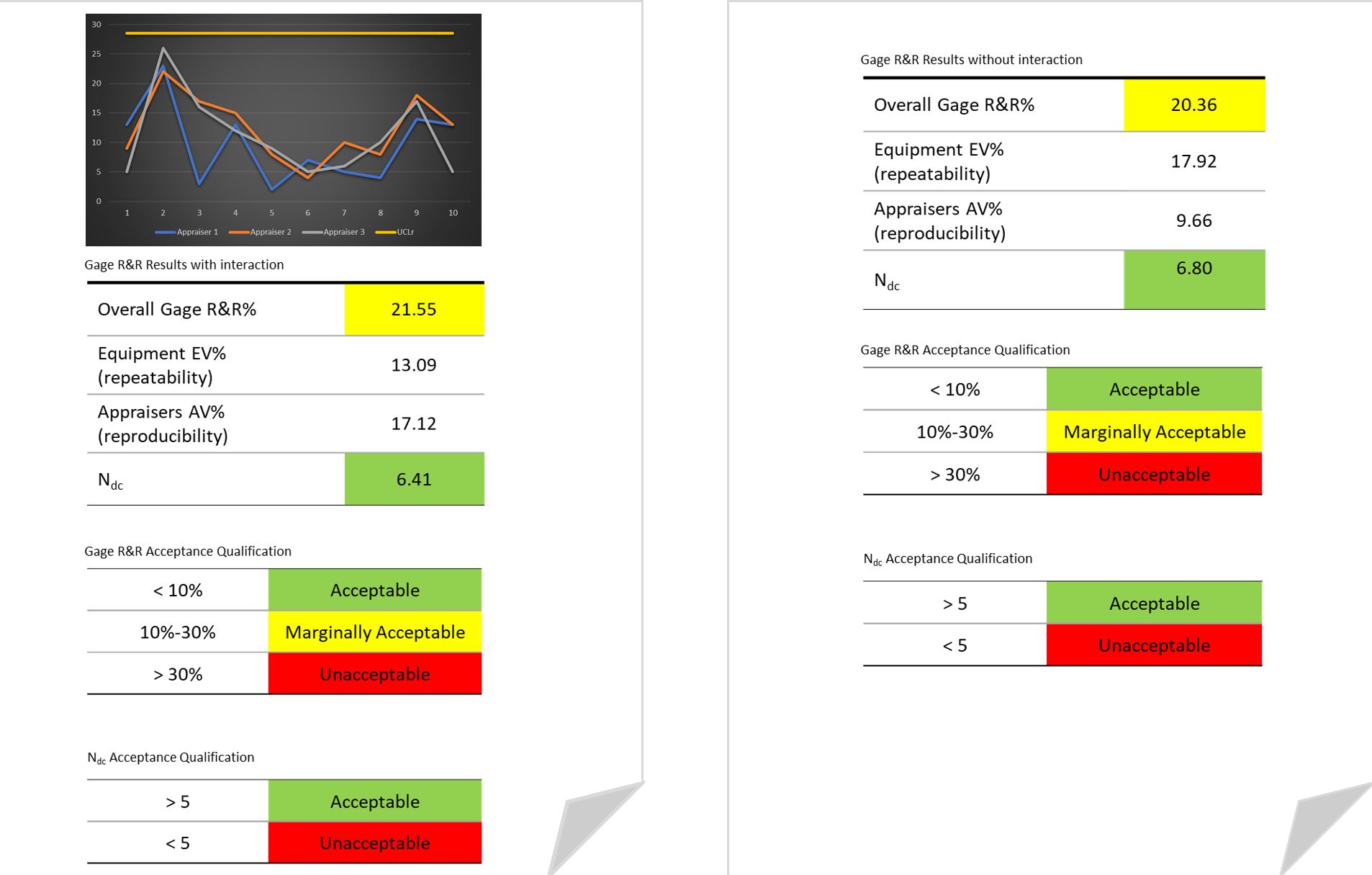
In combining measurement error with product or process variation the resulting value represents the total variation. QIS automated Gage R&R studies allow you to estimate repeatability,
reproducibility and part-to-part variation.
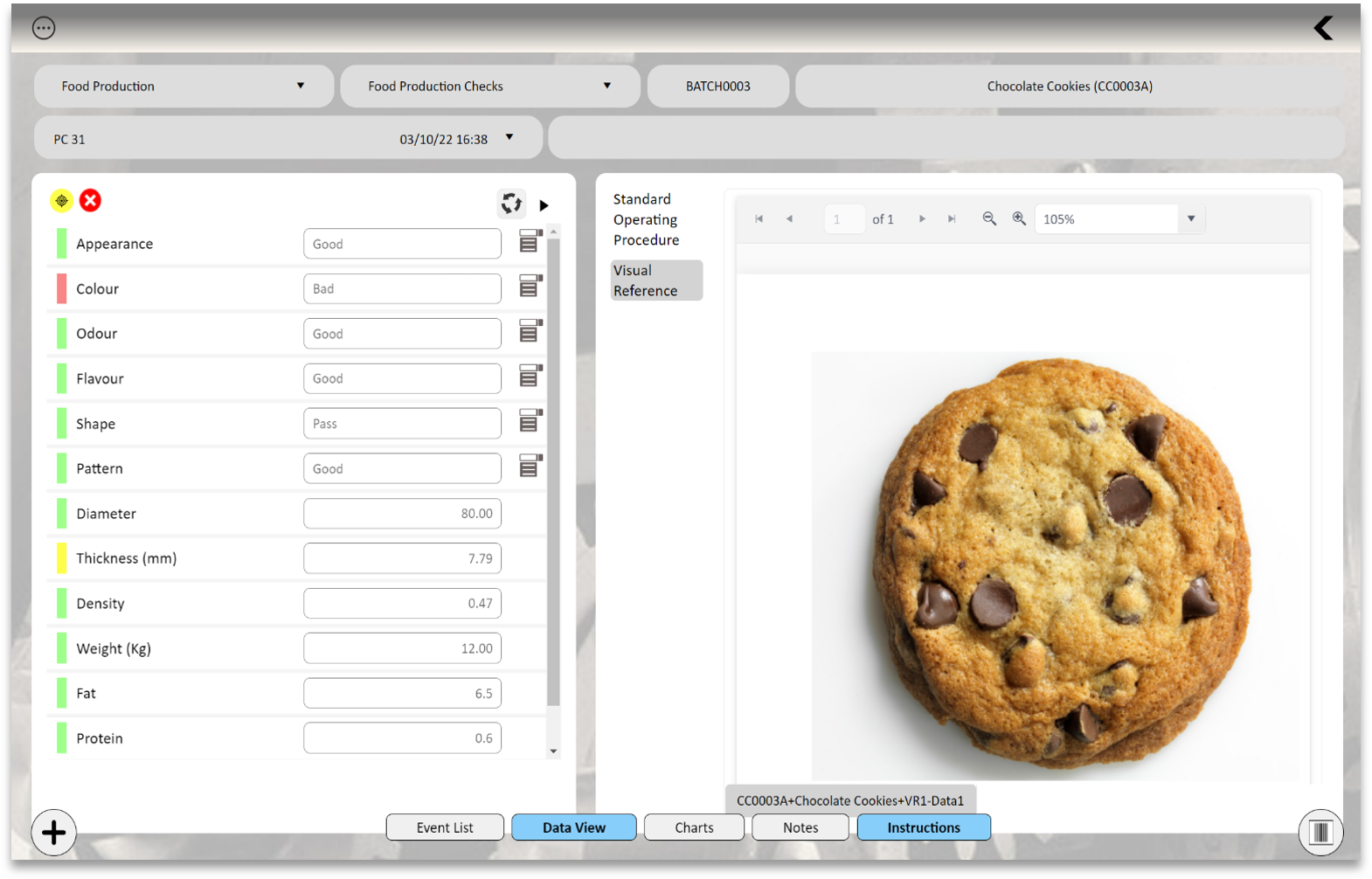
Utilize your central QIS or tablet to review data and associated documentation or test methods. This ensures that those completing the testing have instant access to these vital test methods and standard operating procedures.
Automated Data Capture
Most interface management services run on servers or process networks that regular users do not have access to. The QIS Interface Console provides all authorized users the ability to seamlessly view all logs and any related local settings and the ability to make changes from one location (with the correct rights assigned). For initial set up, re-configuration or any troubleshooting, access to pertinent logs is quick and easy.
Material Tracking& Traceability



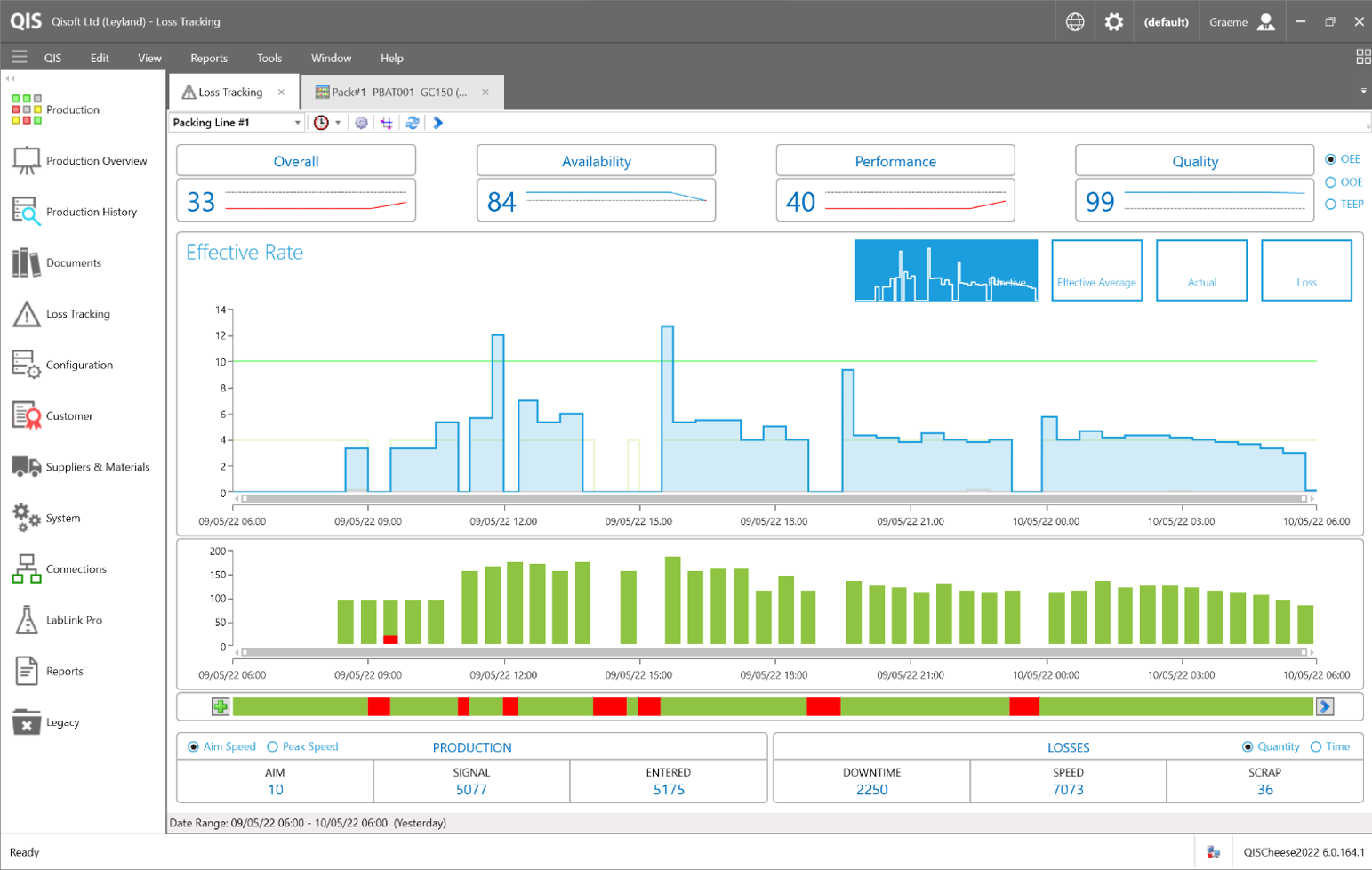
OEE - Production Monitoring
As an integral part of QIS, our asset monitoring and incident-tracking module provides a powerful drill down performance analysis that supports and informs continuous improvement and operational excellence programs. Taking the most common and important sources of manufacturing productivity loss (Quality, Performance and
Availability), and condensing them into an easy-to-use metric.
Helps you tackle inefficiencies within the production proces
Easy to configure and set up for automated incident generation
Powerful and responsive loss tracking dashboards